SPECTROPHOTOMETRY SCAN
SCANNING FOR ELEMENTS…(Minerals, Vitamins and Heavy Metals and More). There has never been a time in history when getting results from blood work was this easy…PLUS…you don’t even need your blood drawn.
A revolutionary non-invasive method for testing Intracellular levels of Minerals and Trace Elements as well as Heavy Metals, using Spectroscopy (Spectrophotometry…a Full Visible Light Spectrum). Intracellular tissue is the destination of what has been bio-accumulated via the blood… which is totally different from what the body keeps in circulation in serum (blood) to keep the body in homeostasis.
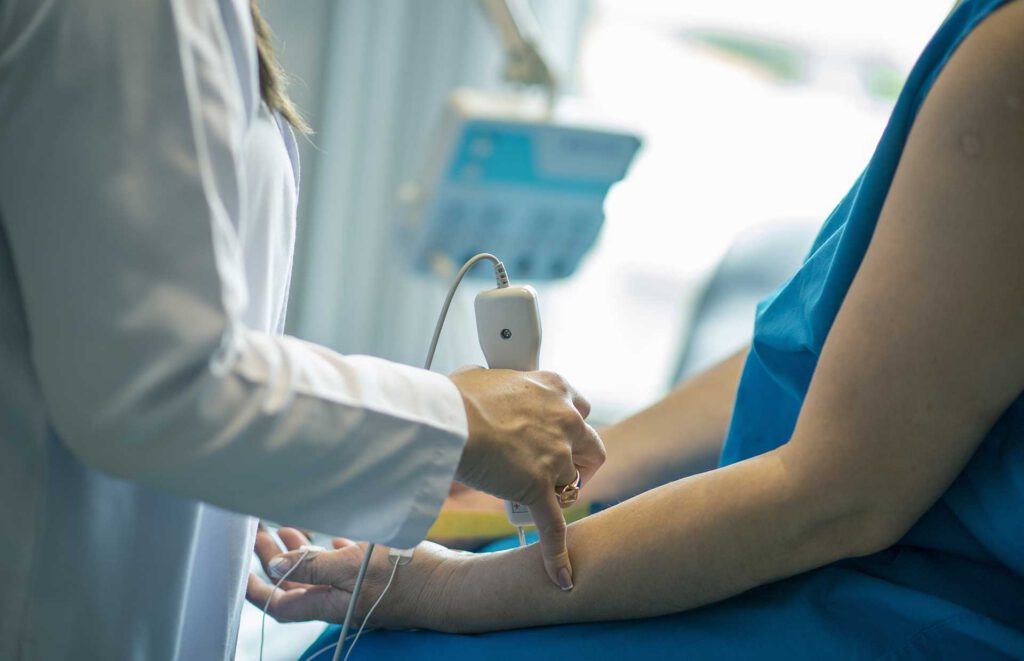
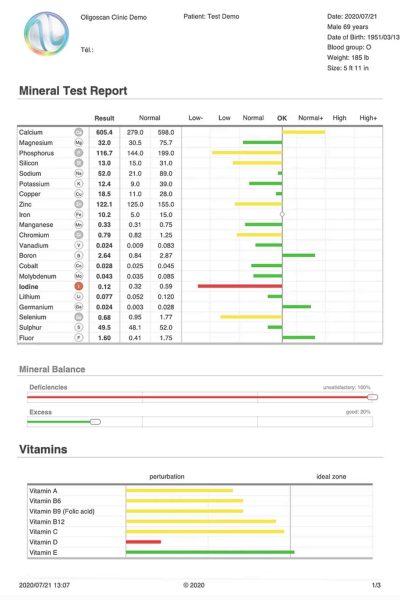
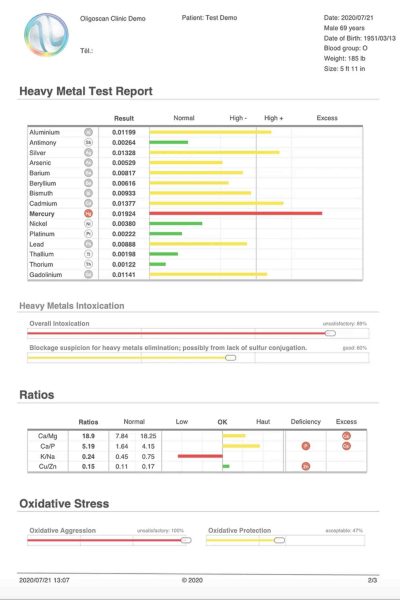
This scan reports on 44 elements—21 Minerals, 16 Heavy Metals, and 7 Vitamins (A, B6, B9, B12, C, D, E). The scan is immediate and non-invasive, measuring the intracellular uptake of the elements in the tissue (including muscle & small capillaries), at a depth of 4mm through the skin using Spectrophotometry.
Information Required
Scan requirements include:
- Date of Birth
- Gender
- Height
- Weight
- Blood Type (to be ascertained by client before the scan)
With Spectrophotometry, the reference range was established by a detailed statistical process utilising Gaussian formulas and functions, a standardised methodology for statistical distribution used in these types of applications over an extended period of time In particular, as intracellular constituents are delivered to cells via the blood, the spectrophotometer enables a clients nutritional status to be measured via two sets of data, as the reference ranges of intracellular component are different from that of the blood.
Therefore, spectrophotometry provides a different view of the uptake of nutrients in the subject. This process is easy, simple and clearer. Gone are the days when how you felt was the only measuring stick, when guesswork guided our health choices and outcomes were left to chance. With today’s technology, we no longer have to shoot in the dark. We now have the tools to gain real, clear and measurable insight into personalized information…though not to be used for diagnostic purposes. You’ve been working hard to detox heavy metals for weeks, months or even years, but how do you truly know it’s working? Answer is, most of the time, you don’t. Progress is often based on how you feel, without measurable proof. But now, there’s a better way.
This scan reveals whether your detox is making a difference or not, thereby helping you track your transformation with the clarity and confidence that is required. The scan is a complementary test that provides a different perspective, not a replacement for medical test.
The Power of Light
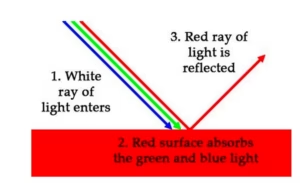
Beer-Lambert’s Law (1760) states that every known compound absorbs, emits, or reflects light (Electromagnetic Spectrum) over a specific range of wavelengths. The more a sample is concentrated, the more it absorbs the specific light applied.
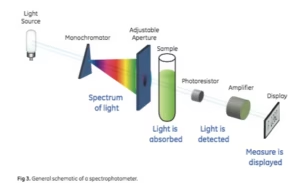
Spectroscopy is all about absorption and reflection.
Every beam of light is quantized (a certain amount of light) into photons, which you can count. As multiple photons of light are projected, there is a consistent and measurable amount of frequencies, creating momentum.
Each Element has its own spectrum of color, or Atomic Emission Spectra, and absorbs and reflects light differently based on the elements’ properties.
When the light hits your skin and starts measuring, it has to penetrate at least 1 cell membrane, otherwise, it can not measure at all. When the light shines into the skin, the momentum changes, in which you can gather and calculate how much of the light was absorbed. The cell membrane is the key difference with Oligoscan.
Advanced Spectroscopy w/ Advanced calculations
Spectroscopy technology is not new and is used across several different industries today such as semiconductors, forensic examination, food-processing, astrology and others.
Otto Heinrich Warburg (1883-1970) was awarded the Nobel Prize in Physiology Medicine in 1931.
“He conducted landmark experiments using Spectrophotometry that proved that the portion of the enzyme that reacts with Oxygen in a cell is identical with the portion of hemoglobin that binds oxygen in the blood. That chemical, called heme, is a porphyrin bonded to iron, and the enzyme containing it, which exists in every cell and makes breathing possible, is known today as cytochrome oxidase.” (The Invisible Rainbow 2017)
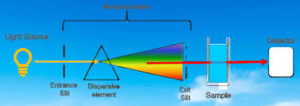
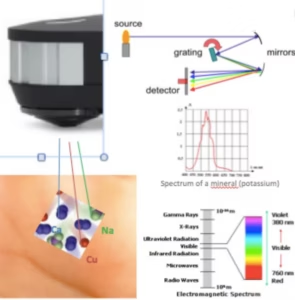
The function of the Spectroscopy is to separate the spectral line of interest from other spectral lines with different wavelengths emitted by the light source (gas filled Tungsten).
A grating is a reflective surface, scored holographically with parallel grooves that are designed for different wavelength regions.
The device is equipped with a diffraction grating with the goal to cover a large wavelength range, which is used in atomic absorption of minerals and metals.
A basic drawing of this technology is shown here. It consists of the diffraction grating (dispersing element), slits, and spherical mirrors.
Atomic absorption of minerals and heavy metals are reflected and detected by their wavelengths E.g. Ca: 422.7nm, K = 766.5 / nm.

Different Tests, Different Reference Ranges
Blood, Hair, Urine, and Spectroscopy each have their own reference ranges developed over time and always in revision.
With Spectrophotometry, the reference range was established by a detailed statistical process utilizing Gaussian formulas and functions over an extended time period. The Gaussian methodology is the standardized method for statistical distribution used in these types of applications.
In particular, as Intracellular constituents are delivered to cells via blood, the spectrophotometer enables a patient’s nutritional status to be measured via two sets of data, as the reference ranges of intracellular components are different from those of blood. Therefore, spectrometry provides a different view of the uptake of nutrients in each individual tested.
The important part for the clinician is that mineral results from each of these four categories will yield different results, because each of the tissues being examined (hair, blood, urine, palmar tissue) is different.
Blood
Represents what is currently in circulation at that moment.
“The Transportation System of nutrients”
- Shows recently circulated Metals, flagging acute intoxication
- Heavy metal levels in the blood are subject to several environmental factors.
- Homeostasis – to preserve life, the body will naturally push or “dump” toxins that enter the bloodstream immediately into the peripheral, fatty, and organ tissues.It does this to stabilize normal bodily functions.
The Amount of Minerals and Trace Elements is kept fairly constant in the blood
- E.g. Muscle Cramp symptoms – Magnesium level in serum & RBC compared to Oligoscan
Hair Mineral Analysis Test
Represents what the patient has excreted in the past 3 months, via epithelial tissue (hair) “The Excretory System”
- 90% of human scalp hair is in the growth phase
- During this time, elements from the follicular cells and their blood supply are incorporated into hair. However, the more toxic metals that are incorporated into organ cells, the less is excreted in hair (i.e Autistic Spectrum Disorder)
- If patient is a poor excreter, as seen with many individuals, notingly with the chronically ill, the toxic elements may appear under-represented in hair tissue compared to Oligoscan
Urine (Provocation) test
- When the body is provoked (e.g. EDTA, DMPS, DMSA), increased metal concentrations are found aka “challenge test”
- Very useful to test excretion capability.
- Note*- chelation agents do not penetrate cell membranes. They determine the amount of metals within the Extracellular space
Spectroscopy
- Intra-cellular Tissue = The Destination
- What has been bio-accumulated, via the blood, into the intra-cellular tissue.
- E.g – The Uptake of minerals vs. what is in circulation
Spectroscopy technology itself is not new and is used across several domains including, chemistry, pharmacy, environment, food-processing, biology, medical/clinical industries, material/chemical engineering, clinical applications, and many others.
Menu